A Pioneering Transplant Surgeon Becomes a Pioneering Transplant Patient

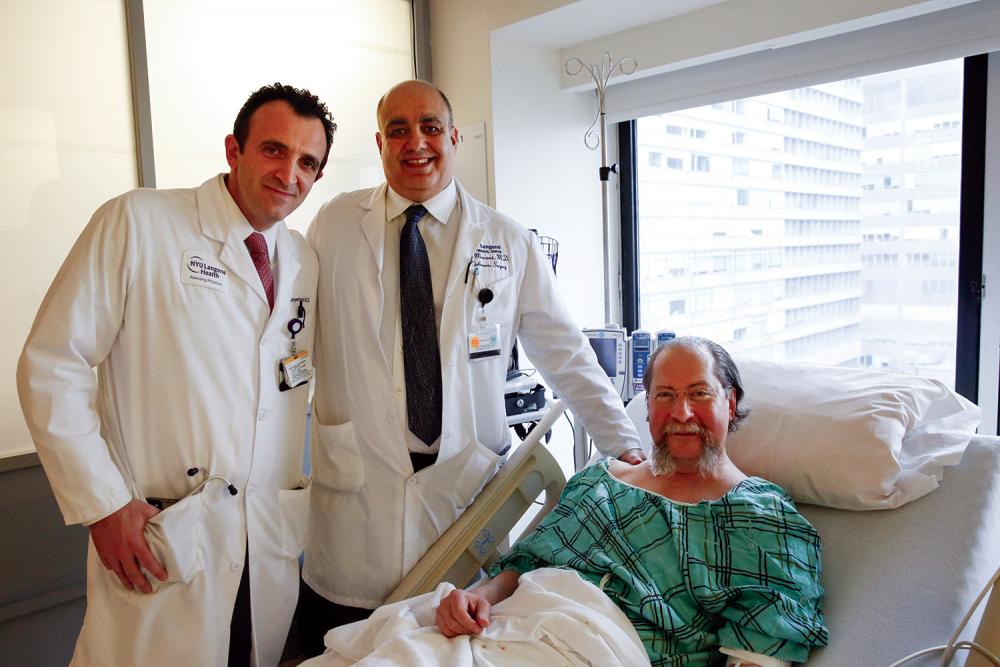
Heart transplant surgeon Dr. Nader Moazami (right) with his boss and patient, fellow transplant surgeon Dr. Robert Montgomery.
Photo: Brad Trent
One night in September 2018, transplant surgeon Dr. Robert Montgomery died. Again. He was sitting on a hotel bed in Matera, Italy, where he had traveled for a medical conference, and was about to get undressed when the palpitations hit. He shouted to his wife, who ran in from the bathroom just as his face hit the stone floor, blood spraying from a gash in his cheek. His heart stopped. Then, after 60 long seconds, the cardiac defibrillator implanted in his chest delivered the shock that brought him back to life.
Dr. Montgomery’s wife, Denyce Graves, struggled to keep her own heart rate in check as she helped him back to bed and pressed a washcloth to his wound. This was not the first time the 58-year-old professor of surgery and director of the NYU Langone Transplant Institute had died and been revived; he’d endured several such episodes during the past 3 decades. But it happened again as the couple waited for an ambulance, and twice more at the tiny local hospital. The staff there, he quickly realized, were ill-equipped to treat his condition. The next morning—against medical advice—he signed himself out and flew home with his wife and a physician friend, who carried syringes full of resuscitation medications supplied by the worried Italian emergency room doctors.
In New York, Dr. Montgomery met with the heads of NYU Langone’s heart transplant team: Nader Moazami, MD, surgical director of heart transplantation and mechanical circulatory support, and professor of cardiothoracic surgery; and Alex Reyentovich, MD, medical director of the heart transplant program and associate professor of medicine. Until recently, neither had known that their boss had a disorder that might make him their patient. Dr. Montgomery suffered from familial dilated cardiomyopathy, a rare, progressive disease of the heart muscle that weakens its pumping ability and causes dangerous arrhythmias. He’d managed to become a renowned kidney transplant surgeon nonetheless, keeping his illness at bay with medications, electronic implants, and sheer willpower. Yet, over the past year, a string of terrifying incidents, of which this was only the latest, had shown that he would need more to survive. “We all agreed he was in serious trouble,” Dr. Moazami recalls.
Although his life-threatening symptoms qualified him for priority status on the transplant list, Dr. Montgomery knew how difficult it could be to get a new heart. His uncommon size (six feet one) and in-demand blood type (O) made him a tough match. Moreover, as with every other organ, there simply weren’t enough to go around. In 2018, some 114,000 Americans needed an organ transplant, but only 36,500 received one; each day, an average of 20 patients died while waiting. About 4,000 people were desperate for a heart, but fewer than half that many hearts became available.
Under Dr. Montgomery’s leadership, however, the Transplant Institute has been pioneering innovative protocols aimed at improving those numbers. Among the most promising is the use of organs infected with hepatitis C (HCV). The deadly virus, carried by an estimated 3.2 million Americans, attacks the liver, typically causing no symptoms until the damage is severe. New antiviral medications, introduced over the past 5 years, have made HCV curable in up to 98 percent of cases, without the grueling side effects of older, less effective drugs. If the virus is detected early, these newer therapies can often eliminate the need for a liver transplant. A growing body of research suggests that they can also make it safe to transplant organs of all types from HCV-positive donors.
Because HCV is most commonly transmitted through shared needles, using these organs may be one way for some good to come out of America’s devastating opioid epidemic, which killed more than 49,000 overdose victims in 2017. Almost 1,500 HCV-positive organs were transplanted last year—triple the number for 2013. Still, they constituted just 4 percent of total transplants, and most were implanted in people who were already HCV positive. (Overdose-death donors as a whole now account for over 13 percent of solid organ transplants, up from 1 percent in 2000.) Only a handful of transplant programs have adopted them, and many potential recipients remain unwilling to accept them. As a result, more than 40 percent of donated HCV-positive organs are discarded.
Dr. Montgomery, for his part, had no hesitation. As he often told his patients, any transplant requires weighing the risks against the potential rewards. “I’ll take any heart you can find,” he told his team. “I don’t care if the donor has a needle in their arm.”
A Family Secret
Robert Montgomery’s comfort with risk emerged early, but it took a tragedy to harness it for a larger purpose. As a young boy growing up in Philadelphia, slow to read and wildly energetic, his heedless behavior got him kicked out of parochial school. “The nuns used to say, ‘Bobby doesn’t believe the rules apply to him,’” he recalls. Then, when he was 13, his father, a mechanical engineer in the aerospace industry, was diagnosed with cardiomyopathy, which doctors mistakenly attributed to a viral infection. His condition deteriorated rapidly, and he suffered three cardiac arrests over the next two years. The last one left him in a chronic vegetative state for several months before his death, at 52.
With his three older brothers away at college and his mother reeling from the loss, 16-year-old Bobby had to grow up quickly. He discovered a passion for medical science, particularly organ transplantation—fueled, in part, by frustration over his father’s inability to obtain a new heart. (At the time, 50 was considered too old to qualify.) During his premed studies at St. Lawrence University, he spent a summer doing aid work in Africa. After graduation, he returned to the continent on a fellowship to study the interface between Western and traditional medicine. The close personal bonds between African healers and their patients would inform his own approach to doctoring. “He’s incredibly caring toward his patients,” says Brigitte Sullivan, executive director of the Transplant Institute.
He went on to medical school at the University of Rochester, and then to Johns Hopkins for his residency. One night during his internship, he received a call from his sister-in-law: one of his older brothers had died of cardiac arrest at 35 while waterskiing. Suspecting that an inherited disorder was the culprit, Dr. Montgomery sent tissue samples from his brother’s and father’s hearts to his old pathology professor, who confirmed his fears. He decided to undergo tests himself. On the treadmill, his heartbeat went wild, and he almost passed out.
So, in 1989, Dr. Montgomery became the first practicing surgeon in the world to receive an implantable cardiac defibrillator—a device that had just been developed at Johns Hopkins. In those days, the procedure required open-heart surgery. A generator the size of a soda can was placed in his abdomen, powering a pair of large capacitors attached to the outside of his heart. The apparatus made it painful to wear a belt—but worse, it threatened to dash his dreams of becoming a transplant surgeon. (He’d decided to focus on abdominal organs, because heart surgeons of that era typically performed transplants only as a sideline.) “We’d like you to take a few years off and do some research,” his chair of surgery told him. “Let’s see how this thing progresses.”
With his pregnant first wife, Dr. Montgomery set off for the University of Oxford to pursue a doctorate in molecular immunology. Soon after arriving, he heard a crash outside his apartment. Running to the street, he found a car turned upside-down with a baby strapped inside and a woman screaming on the pavement. He climbed in through the window and extricated the baby. Then, he felt a blow like a baseball bat slamming against his chest as his defibrillator discharged, triggered by his racing heartbeat.
At that moment, he realized that he could never resume his surgical career, in which steadiness under pressure was the most basic job requirement, unless he learned to control his response to stress. “I had to completely remodel my brain and not allow my body to react,” he explains. Like a self-taught Zen master, he began to monitor his emotions and his flow of adrenaline, forcing himself to grow calmer as a situation grew more challenging.
In 1992, having earned his doctorate, Dr. Montgomery returned to Johns Hopkins and was cleared to finish his training in general surgery and later in multiorgan transplantation. During his surgical fellowship, he helped develop the first laparoscopic kidney procurement technique. The innovation made recovery easier for donors, facilitating donations of live-donor kidneys, which last much longer than those from cadavers. By 2003, he’d become director of the transplant program at Johns Hopkins, where he gained a reputation as a trailblazer in efforts to get more organs to people who needed them.
One of his innovations was the “domino” kidney transplant, which occurs when several people who need transplants have friends or relatives who are willing to donate but aren’t compatible; a chain of surgeries is arranged in which each would-be donor is matched with a compatible recipient, with “altruistic” donors, who are willing to give a kidney to anyone, filling in the gaps. An eight-way swap in 2010 landed Dr. Montgomery in the Guinness Book of World Records. He also helped develop a protocol combining kidney and bone marrow transplants to prevent rejection of donor organs in immune-incompatible patients and eliminate the need for immunosuppressive therapy. He was also among the first transplant surgeons to promote the idea of using HCV-positive organs, after the new generation of antivirals proved capable of quickly clearing the virus.
Recruited to NYU Langone in March 2016 to found the Transplant Institute, Dr. Montgomery (along with Brigitte Sullivan, who accompanied him from Johns Hopkins as executive director) brought together transplant specialists from all disciplines under one roof, enabling them to pool skills and insights without the territorial barriers found at many institutions. A slew of new hires from across the country enhanced the institute’s capabilities. The kidney transplant program soon added a pediatric group. Dr. Moazami, who formerly headed the heart transplant program at the Cleveland Clinic, where he helped develop a range of novel surgical and device-based approaches to heart failure, led NYU Langone’s first heart transplant procedure in January 2018. The institute’s first lung transplant followed just weeks later, and its first pancreas transplant some months after that.
Dr. Montgomery also launched several initiatives to identify potential donors more swiftly and to ensure that potentially viable organs often rejected for transplantation didn’t go to waste (including those infected with HCV or HIV). Within 2 years of his arrival, NYU Langone had increased the number of organ transplants from fewer than 50 to more than 280. Its heart transplant rate, or the number of transplants each year divided by the number of patients on the waiting list, was five times the average for the New York region. Its wait times for all organs was the shortest—an average of 36.3 days for a heart, versus 66.1 across the metropolitan area.
However, when Dr. Montgomery went on the waiting list in September 2018, those statistics were scant comfort. No one could say whether he would survive long enough to benefit from his own breakthroughs.
Defining the Limits
As Dr. Montgomery waited in a patient room at the Helen L. and Martin S. Kimmel Pavilion, tethered to vital signs monitors, he had time to reflect on his vulnerability—something he had largely avoided doing during the course of his extraordinarily active life. From the day he was diagnosed, he’d been determined to prove that his illness could not defeat him. Yet, he’d never known when the next crisis would hit. His antiarrhythmia medication had ruined his thyroid. His defibrillator’s batteries needed replacing every few years, requiring major surgery. When the leads wore out, they began triggering shocks whenever he lifted his kids. He’d eventually gotten a smaller, more modern device installed, but much of the old hardware remained in his chest.
To defy his disease, Dr. Montgomery had taken up strenuous pastimes in exotic places—bow hunting in Argentina, fly-fishing in Tanzania. Even there, however, escape was only provisional. In 2012, researchers supported by his family’s charitable foundation had isolated the genetic mutation responsible for his type of cardiomyopathy. Dr. Montgomery’s older son and daughter, as well as his late brother’s two daughters, proved to have it, and all were implanted with defibrillators. (Another brother had already undergone a heart transplant and is still healthy at 62; the remaining brother is not affected.) Later that year, Dr. Montgomery was on a mountain in the Andes when his defibrillator went off, knocking him face down in the snow. His son helped him hike to safety. From that point on, he rode in the jeep instead of climbing, but he refused to give up his adventurous trips. “I had to set an example for the rest of the family,” he says. “I wanted them to know you could have this and still live a full life.”
But his resistance had begun to fail. In 2017, back in Argentina, he contracted a drug-resistant strain of pneumonia. Near death, he was placed on a ventilator. Luis F. Angel, MD, professor of medicine and of cardiothoracic surgery, and medical director of the lung transplant program, flew down and rushed him to NYU Langone, where he spent weeks recovering. That October, he went into cardiac arrest while watching a Broadway show with his family. This time, his implanted device initially failed, and he needed prolonged CPR before he came back to life. Afterward, his defibrillator was modified to administer a stronger shock, and he was fitted with a pacemaker. Then came the incident in Italy.
Now, here he was, yearning for a reprieve like so many of his patients over the decades. In the future, he thought, he could tell them truthfully that he knew how they felt.
Boss, Friend, Patient
The hoped-for call came at 4:00AM on September 20, just five days after he was admitted to NYU Langone. “We have a donor,” said Dr. Moazami. “Heroin overdose, in their 20s, hepatitis C+.” Dr. Montgomery didn’t pause. “Let’s go,” he said. Within a few hours, Zachary N. Kon, MD, assistant professor of cardiothoracic surgery and surgical director of the lung transplant program, was in a neighboring state, procuring the donor’s heart and preparing it for transport. Meanwhile, Dr. Moazami was in an operating room at NYU Langone with Deane E. Smith, MD, assistant professor of cardiothoracic surgery and associate director of heart transplant and mechanical circulatory support, opening Dr. Montgomery’s chest. It took an hour to remove the tangle of wires and capacitors that remained from his old implants. Around 2:00PM, when Dr. Kon’s chartered plane landed at Teterboro Airport, the two surgeons placed their patient on a heart–lung bypass machine and began to carefully remove his heart.
By the time Dr. Kon walked in, with the donor organ in a wheeled cooler, his colleagues were ready to begin the transplant. For Dr. Moazami, the fact that the man under his knife was not only his boss but also his friend was not a particular cause for anxiety. “Every patient is a VIP in my mind,” he says. “They’re all someone’s father, mother, husband, wife, son, daughter. And I was confident in our team.” It also helped that he had smuggled himself out of Iran as a teenager, made it through college and medical training on his own, and held the hearts of hundreds of other men and women in his hands. Like Dr. Montgomery, he had trained himself to respond to stress with intense calm.
At 2:46PM, Dr. Moazami lifted the new heart—healthy and strong, despite the virus that lurked in its cells—and placed it in the recipient’s thoracic cavity. At 6:09PM, nearly seven hours after the surgery began, he and Dr. Smith were closing up the incision. “Good job, everyone,” Dr. Moazami said. “Thanks for everything you’ve done.” Afterward, one of the nurses approached him with tears in her eyes. “Dr. Montgomery did my daughter’s kidney transplant,” she said. “I hope he’ll be all right.”
Young at Heart
He was all right. Although his new heart had some rhythm problems at first, they were resolved with medication. Dr. Montgomery went home 10 days later. By then, he’d tested positive for HCV, as expected, and a course of medication had cleared the virus. Less than a week after his discharge, he returned to work halftime. “You can’t slow him down,” says Dr. Reyentovich, who had suggested that he wait longer. Soon, Dr. Montgomery was back to his usual long hours at the office. He even began doing cardio exercises as part of his recovery program at Rusk Rehabilitation. “For the first time in years, I can get on a treadmill and run,” he marvels.
However, eight months after his surgery, Dr. Montgomery’s life has not quite returned to normal. Like most transplant recipients, he takes a triple-drug immunosuppressant cocktail to ward off rejection; a dozen more medications each day to prevent infections, to which the immunosuppressants make him vulnerable; and pills to prevent coronary artery disease in his new heart. He’ll be on some version of this regimen, with periodic biopsies to make sure it’s working, forever. (The median survival for a transplanted heart is at least 12 years.) One of the immunosuppressants causes a hand tremor—a minor annoyance for most patients, but not for a surgeon. The effect typically wears off after a few months, but if it doesn’t, he plans to switch to a different drug.
Still, Dr. Montgomery is eager to pick up the scalpel again as soon as safety allows. He’s grateful for the gift in his chest, provided by a grieving family, and he’s glad for the chance to prove that the risk he took in accepting it can benefit many people other than himself. Everyone who receives an HCV-positive organ at NYU Langone (about half of all heart recipients) does so as part of a clinical trial—one that, so far, has shown striking results. “No transplant patient has failed to respond to the hepatitis C therapy we’ve given,” says Ira M. Jacobson, MD, professor of medicine and director of hepatology, who led pivotal early studies of the new antivirals and helped design the Transplant Institute’s treatment protocols.
“The virus is suppressed very quickly. To me, it’s a thrill to do research that can have an impact around the world,” Dr. Montgomery says.