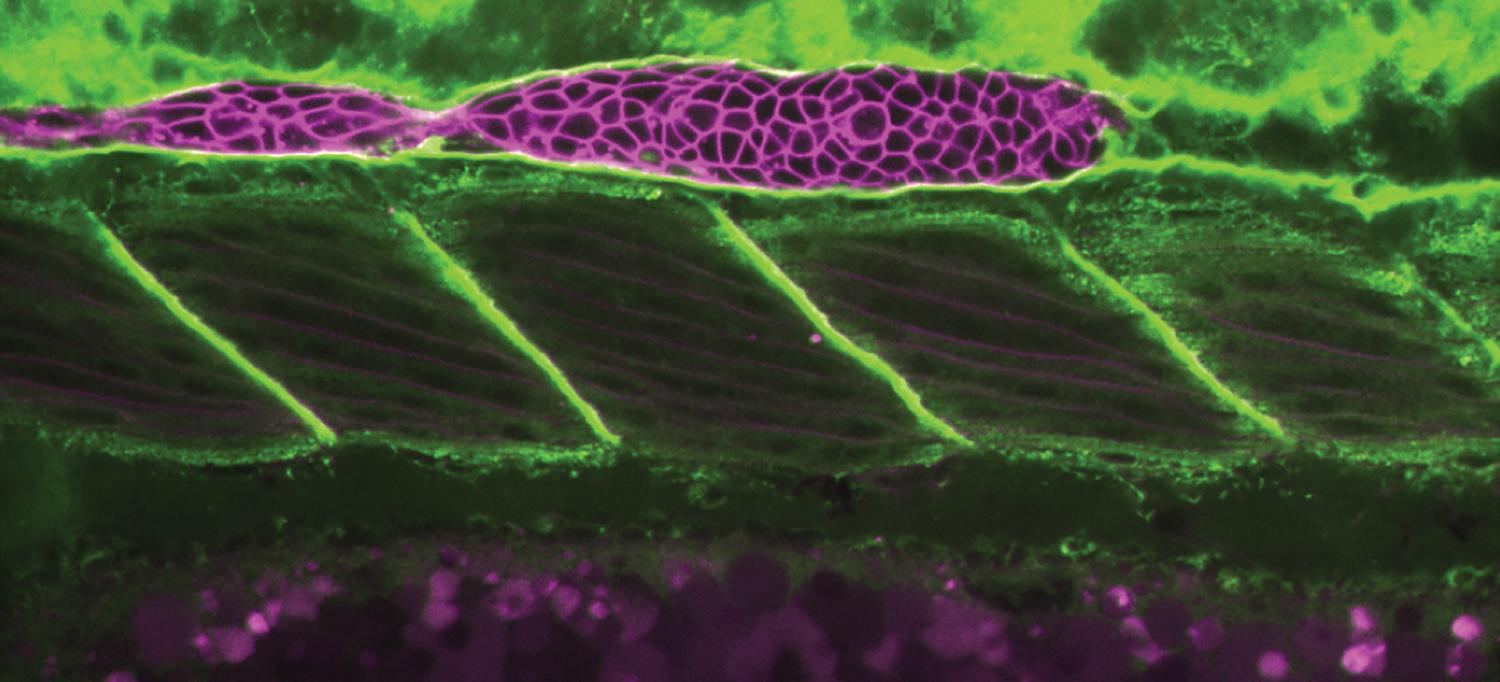
A moving cell group called the primordium, in purple, moving to the right along the tissue it grips, the basement membrane, in green.
Photo: Knaut et al.; Nature Cell Biology
Cells push and pull on surrounding tissue to move in groups as they form organs in an embryo, track down invading bacteria, and become cancerous and spread.
Published online in Nature Cell Biology on February 14, a new study has found that in a living embryo, the back ends of moving cell groups push the group forward. This runs contrary to previous findings, where cell groups grown in dishes of nutrients (cultures) pulled themselves forward with their front edges.
Led by researchers from NYU Grossman School of Medicine and NYU Courant Institute of Mathematical Sciences, the study used a new technique to measure the forces applied by a cell group as it moved along a “road-like” tissue membrane and into place in a developing animal. Specifically, the study found for the first time in an animal tissue that proteins called integrins on the surfaces of the cells at the rear attach in greater numbers to the membrane as they move along, and exert more force in one direction, than the cells in the group’s front. The integrin clusters (focal adhesions) observed in the embryo were smaller than those seen in culture studies, and broke down faster.
Confirmation of such mechanistic details in living tissue has important implications, say the researchers, as many cancers spread in cell groups and may use the newfound “rear engine propulsion.”
“Our results clarify how cell groups that will become organs move into place, and reaffirm that cells behave differently when removed from their natural environments,” says senior study author Holger Knaut, PhD, associate professor in the Department of Cell Biology at NYU Langone Health.
How the Study Was Conducted
The study results are based on mechanisms of cell movement established by past studies. For instance, a protein called actin is known to form the protein “skeleton” of cells, with actin chains able to grow in a certain direction, and apply force that changes a cell’s shape. Integrins, proteins built into outer cell membranes, interact both with actin networks and with proteins outside of cells. These and other proteins form a system that a cell uses to briefly attach to and “roll along” a basement membrane, a pliable mesh of proteins and sugars. What was unknown going into the current study was how tissues in living animals apply force in groups to generate this motion.
The new study examined cell group motion in a zebrafish embryo, a major model in the study of development because it shares many cellular mechanisms with human cells and because zebrafish embryos develop externally, such that each stage in development can be directly observed using high-powered microscopes. In this way, the team tracked the movement of the primordium—a tissue made up of about 140 cells—as it migrated during development from behind the ear to the tip of the zebrafish tail, where it matured into an organ that senses water flow.
“In the first study of its kind, we combined advanced microscopy with automated, high-throughput computational modeling to measure cellular forces in living organisms,” says co-corresponding author Daniele Panozzo, PhD, an associate professor at NYU Courant Institute of Mathematical Sciences.
Using “bleached” dots on the basement membrane to measure shape changes (deformations) on a minute scale and a new software called embryogram to calculate how far the dots move as the primordium “grips” the membrane, the researchers determined how much the cells pulled and pushed on the membrane, “like a tire on pavement.” The effect is much like the high school physics experiment where students draw two dots on a rubber band and calculate the force applied as they stretch the band by measuring the change in distance between the dots.
With these tools in hand, the team showed that the primordium cells link the force-generating actin–myosin network at the back end of the moving group through integrin clusters on the side closest to the basement membrane. The team theorizes that cells attached to the membrane toward the back push on the cells in front of them to move the entire group. The researchers also gained new insights on an established mechanism where cells have surface proteins that let them “sense” and follow a guidance cue called a chemokine from low concentration to high concentration. The new study found, however, that cells toward the back end of the primordium sense the chemokine gradient more strongly.
Interestingly, the study found that the primordium moved in a “continuous breaststroke” by pushing the basement membrane downward, sideways, and backward, much like the arms of a swimmer. The authors do not know why this is, but they speculate that it is the most efficient way to move forward. They note that banana slugs also use the rear edge of the “foot” they apply to the ground, suggesting that evolution favors rear engine propulsions because they are most efficient at different size scales.
The study suggests that group cell movement has the potential to be harnessed to stop cancer spread, perhaps by designing treatments that block the action of integrins, say the authors. Integrin inhibitors have been tested as drugs for cardiovascular and autoimmune disease in clinical trials, but their use against cancer spread has been limited by the need for a better understanding of the mechanisms.
Along with Dr. Knaut and Dr. Panozzo, study authors were Naoya Yamaguchi, Ziyi Zhang, and Teseo Schneider from NYU, and Biran Wang from Memorial Sloan Kettering Cancer Center. The work was supported by National Institutes of Health grant NS102322, NYSTEM fellowship C322560GG, American Heart Association fellowship 20PRE35180164, and National Science Foundation grants 1652515, IIS-1320635 (D.N.), OAC-1835712, OIA-1937043, CHS-1908767, and CHS-1901091, as well as by gifts from Adobe Research, nTopology, and Advanced Micro Devices, Inc. The investigators have no disclosed interests relating to these companies, and the gifts are being managed in accordance with NYU Langone Health policies and procedures. Questions regarding these interests may be directed to cimu.disclosures@nyulangone.org.
Media Inquiries
Greg Williams
Phone: 212-404-3500
gregory.williams@nyulangone.org